Current Takara Bio one-step RT-qPCR kits being used for COVID-19 detection
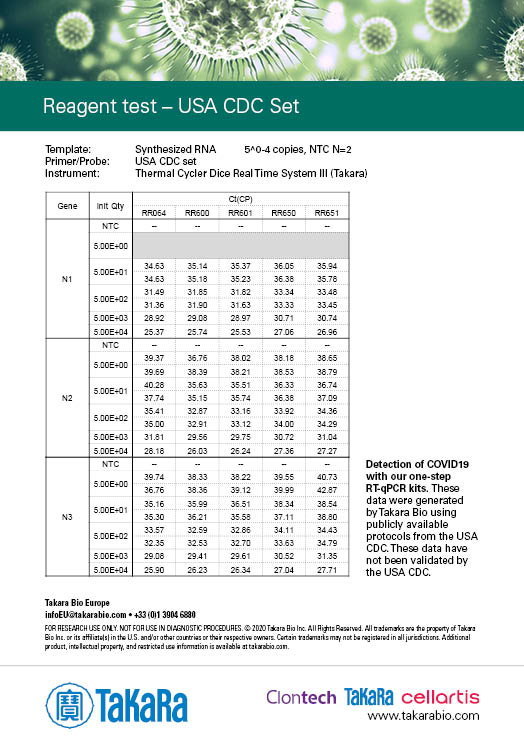
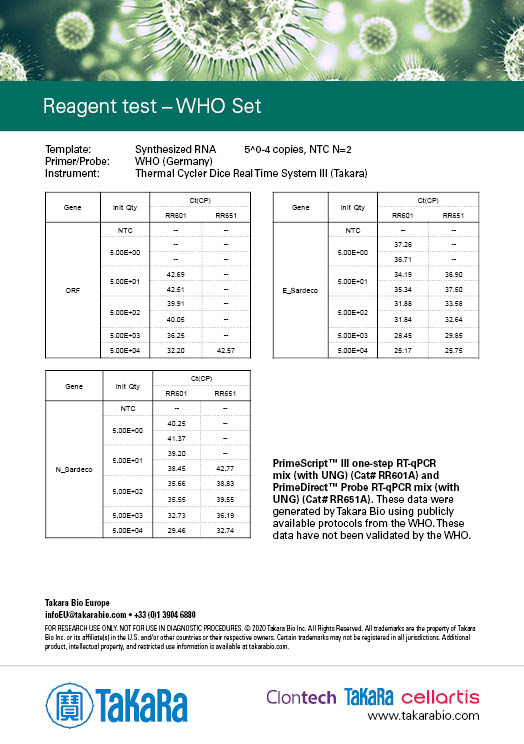
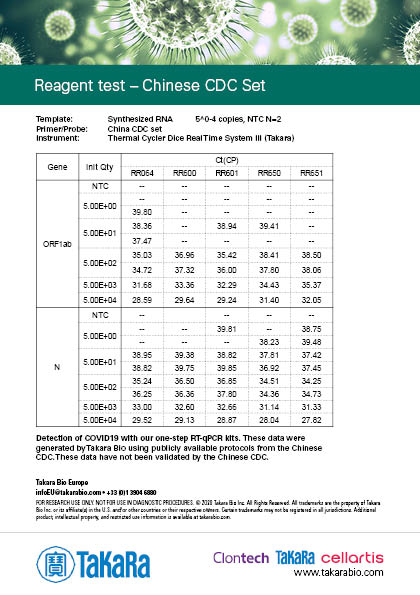
Takara Bio has developed one-step probe RT-qPCR kits that are currently being used in the Chinese CDC and the Japanese NIID protocols for COVID-19 detection:
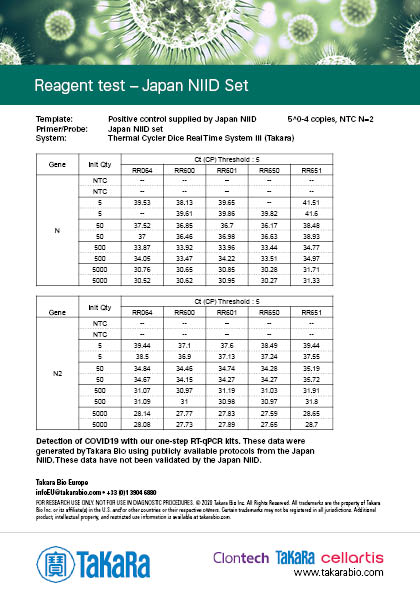
Japanese National Institute for Infectious Diseases (NIID) Protocol
Japanese NIID Protocol translation »
Featured product:
PrimeDirect™ Probe RT-qPCR Mix
Faster, simpler virus detection directly from biological samples
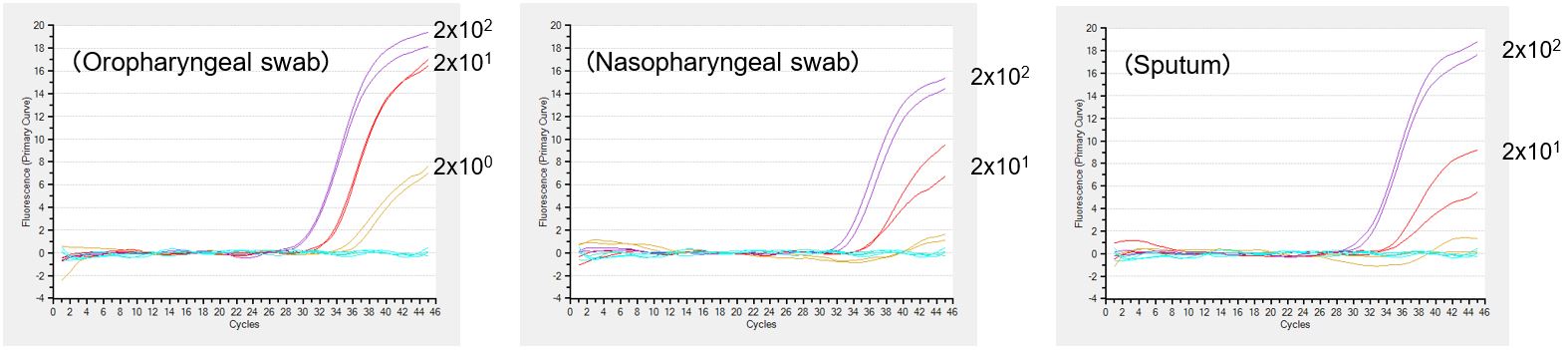
Detection of inactivated influenza A virus H1N1
Inactivated influenza A virus was spiked into oropharyngeal swab samples, nasopharyngeal swab samples and sputum samples at the concentration of 2 x 100-102 copies/µl, and the detection was performed for each sample type with PrimeDirect™ Probe RT-qPCR Mix. Results were generated by Takara Bio.
| Test material | Instructions |
| Oropharyngeal swab | Soak the sampled oropharyngeal swab into 200µL PBS buffer, shake for 5 seconds and add 1µl of supernatant to reaction |
| Nasopharyngeal swab | Soak the sampled nasopharyngeal swab in 200µL PBS buffer, shake for 5 seconds and add 1µl of supernatant to reaction |
| Sputum | Directly add 1µl of supernatant to reaction |
| Reagent | Volume |
| PrimeDirect Probe RT-qPCR Mix (2X) | 12.5 |
| Forward Primer (10µM) | 0.5 |
| Reverse Primer (10µM) | 0.5 |
| TaKaRa qPCR Probe (10µM) | 0.5 |
| Test Sample | 1 |
| Influenza A virus H1N1 (2×100~102 Copies/µL) | 1 |
| RNase Free H2O | 9 |
| Total | 25 |
PrimeDirect™ Probe RT-qPCR Mix enabled the detection of viral RNA at 2 copies/µL from oropharyngeal swab samples. The detection sensitivity from nasopharyngeal swab samples and sputum samples was 20 copies/μL. This product is for research use only. It is not intended for therapeutic or diagnostic procedures.