Innovative tools to accelerate vaccine research and development
The recent emergence of the lethal coronavirus, SARS-CoV-2 (COVID-19), has led to an urgent need to rapidly develop a vaccine against this novel virus. Takara Bio offers an array of innovative technologies to support researchers and clinicians in their efforts to quickly develop a suitable vaccine, using reverse vaccinology workflows:
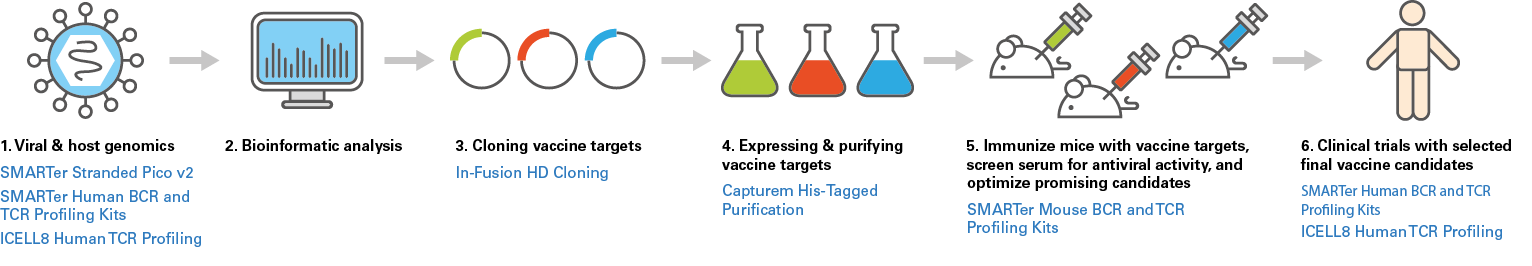
Reverse vaccinology workflow
1. Viral & host genomics
Whole-genome sequencing of the SARS-CoV-2 virus can provide valuable information on potential antigenic epitopes that could trigger an immune response. Indeed, Takara Bio's SMARTer® Stranded Total RNA-Seq Kit v2 - Pico Input Mammalian has been used in China to sequence SARS-CoV-2, providing full-length viral genome sequence.
In addition, BCR and TCR immunoprofiling of COVID-19 infected or recovered individuals via RNA-seq can provide additional information to further fuel the process of antigen and vaccine target identification. Our highly sensitive SMARTer Human BCR and TCR profiling kits can be used to monitor these changes in the BCR-TCR clonotype repertoires of infected or recovered COVID-19 patients.
For automated, high-throughput, human TCR profiling at the single-cell level, SMARTer TCR kits have been adapted for the ICELL8® single-cell system. The appropriate sequencing data analysis pipeline was developed to allow the TCR profiling of single T-cells. Furthermore, it is possible to access the gene expression of the selected T-cell.

2. Bioinformatic analysis
Bioinformatic analysis to reveal SARS-CoV-2 antigens and selection of vaccine targets (viral surface receptors).

3. Cloning vaccine targets
Researchers can speed up viral receptor and vaccine construct generation, including HTP cloning, using our In-Fusion® HD Cloning technology:
- Highly efficient—over 95% cloning efficiency for inserts ranging from 0.5 to 15 kb
- Sequence independent—clone ANY insert, into ANY vector, at ANY locus
- Seamless construction—no extra base pairs (or scar sequences) added to inserts
- Versatile—multiple insert cloning and site directed mutagenesis in a single step
Indeed, a number of publications have already utilized In-Fusion® HD Cloning technology within their workflows to study and develop vaccines against coronaviruses, including SARS-CoV-2:
- Letko, M. et al., Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat Microbiol 5, 562–569 (2020).
- Kato H. et al., Development of a recombinant replication-deficient rabies virus-based bivalent-vaccine against MERS-CoV and rabies virus and its humoral immunogenicity in mice. PLoS ONE 14(10): e0223684. (2019)
- Terada Y. et al., MERS coronavirus nsp1 participates in an efficient propagation through a specific interaction with viral RNA. Virology 511:95-105. (2017)

4. Expressing & purifying vaccine targets
Researchers can then rapidly purify His-tagged vaccine candidate proteins using our revolutionary Capturem high-capacity membrane-based purification technology:
- Fast, convenient workflow—5 to 30 minute room-temperature protocol using convenient spin columns/plates/filtration devices featuring Capturem nickel-functionalized membranes
- High purity—small bed volumes trap fewer contaminants
- Compatible with wide range of additives—including EDTA, DTT, BME, glycerol, TCEP, etc.; see compatibility table)
- Versatile—easy purification from mammalian/bacterial cell lysates and supernatants
Selected publications have already utilized our Capturem His technology within their vaccine development workflows:
- Do V.T. et al. Recombinant adenovirus carrying a core neutralizing epitope of porcine epidemic diarrhea virus and heat-labile enterotoxin B of Escherichia coli as a mucosal vaccine. Arch Virol 165, 609–618 (2020)
- Martínez-Hernández S.L. et al. An anti-amoebic vaccine: generation of the recombinant antigen LC3 from Entamoeba histolytica linked to mutated exotoxin A (PEΔIII) via the Pichia pastoris system. Biotechnol Lett 39, 1149–1157 (2017).

5. Immunize mice with vaccine targets, screen serum for antiviral activity, & optimize promising candidates
Takara Bio's SMARTer Mouse BCR and TCR profiling Kits can be used to speed up the process of immune serum screening for antiviral activity of target vaccines. Promising vaccine candidates are then further optimized prior to going into clinical trials.

6. Clinical trials with selected final vaccine candidates
Monitoring the immune response is key to assessing the effectiveness of the final vaccine candidates. BCR and TCR repertoire profiling by NGS allows precise monitoring of clonotype changes and identification of vaccine-specific clonotypes.
Moreover, the ICELL8® single-cell system in conjunction with our adapted SMARTer TCR kit chemistry, can be utilized here to automate and perform high-throughput TCR profiling at the single-cell level.
Explore Takara Bio's products to accelerate vaccine research and development:
Contact us today to discuss your COVID-19 vaccine development projects
E-mail our Tech Support team at [email protected]. We'd happy to discuss your projects.